Research Symposium
26th annual Undergraduate Research Symposium, April 1, 2026
Amanda Altree Poster Session 3: 1:45 pm - 2:45 pm / Poster #165
BIO
Hi! I'm a junior biochemistry student from Fort Lauderdale, Florida. I'm passionate about biochemistry and our environment. In my free time I love to travel, bake, and visit state parks.
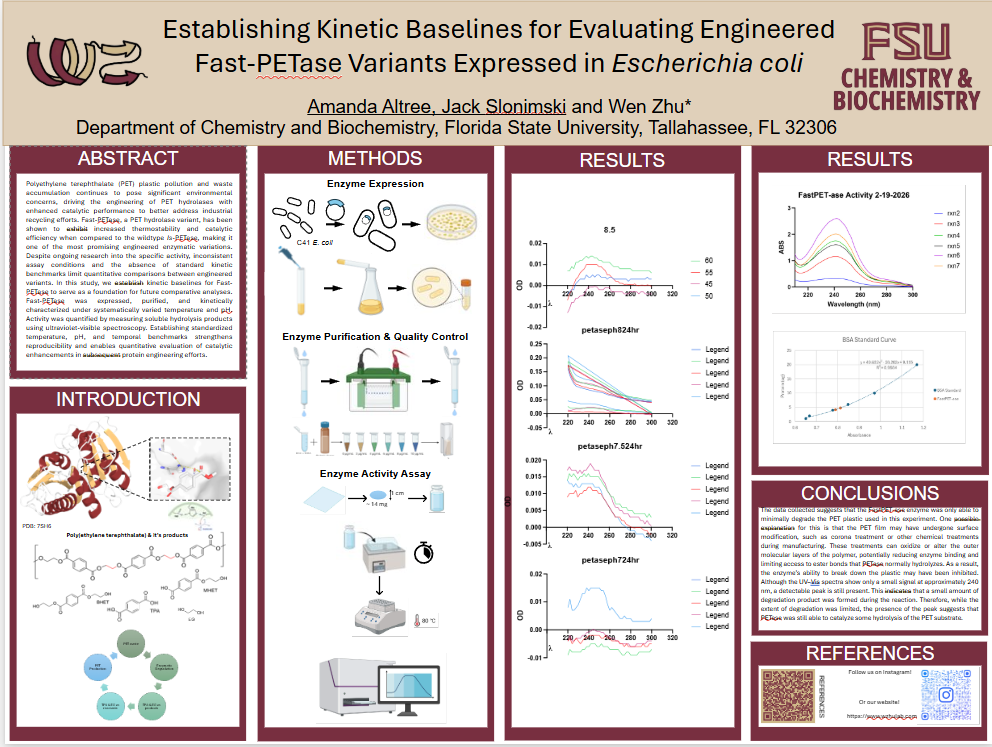
Establishing Kinetic Baselines for Evaluating Engineered Fast-PETase Variants Expressed in Escherichia coli
Authors: Amanda Altree, Wen ZhuStudent Major: Biochemistry
Mentor: Wen Zhu
Mentor's Department: Chemistry & Biochemistry Mentor's College: Purdue University, Cardiff University, University of California Berkeley Co-Presenters: Jack Slonimski
Abstract
Polyethylene terephthalate (PET) plastic pollution and waste accumulation continues to pose significant environmental concerns, driving the engineering of PET hydrolases with enhanced catalytic performance to better address industrial recycling efforts. Fast-PETase, a PET hydrolase variant, has been shown to exhibit increased thermostability and catalytic efficiency when compared to the wildtype Is-PETase, making it one of the most promising engineered enzymatic variations. Despite ongoing research into the specific activity, inconsistent assay conditions and the absence of standard kinetic benchmarks limit quantitative comparisons between engineered variants. In this study, we establish kinetic baselines for Fast-PETase to serve as a foundation for future comparative analyses. Fast-PETase was expressed, purified, and kinetically characterized under systematically varied temperature and pH. Activity was quantified by measuring soluble hydrolysis products using ultraviolet-visible spectroscopy. Establishing standardized temperature, pH, and temporal benchmarks strengthens reproducibility and enables quantitative evaluation of catalytic enhancements in subsequent protein engineering efforts.
Keywords: Biochemistry, PET-ase, FastPET-ase, Enzyme, Plastic