Research Symposium
26th annual Undergraduate Research Symposium, April 1, 2026
Isabelle Rodriguez Poster Session 2: 10:45 am - 11:45 am / Poster #300

BIO
Isabelle Rodriguez is a 2nd year student majoring in Pre Clinical Professions on the Pre Med path working in Solis lab with her mentor Dr.Tigro.
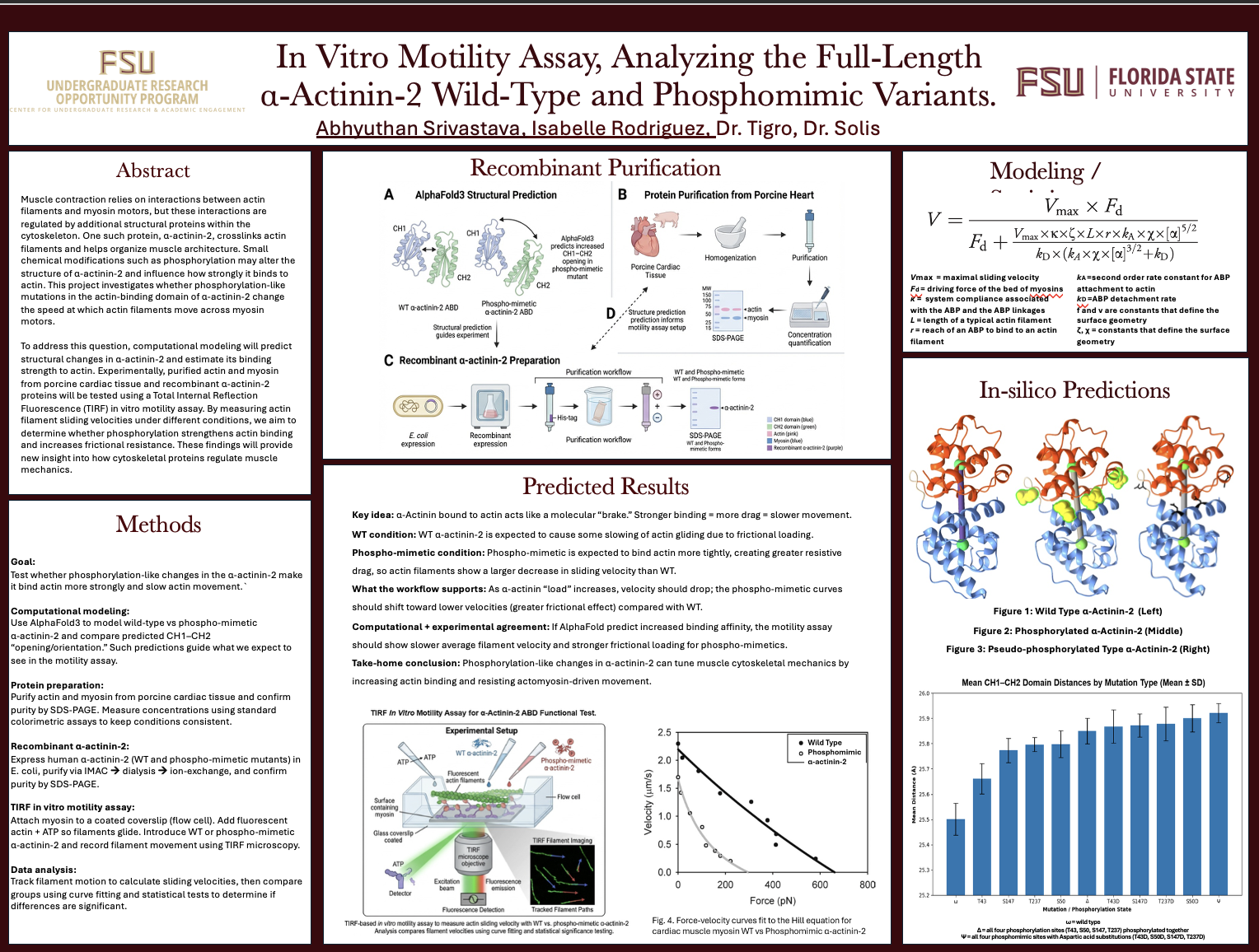
In Vitro Motility Assay, Analyzing the Full-Length α-Actinin-2 Wild-Type and Phosphomimic Variants.
Authors: Isabelle Rodriguez, Helene TigroStudent Major: IMS Pre Clinical Professions
Mentor: Helene Tigro
Mentor's Department: Health, Nutrition, & Food Sci Mentor's College: Anne's Co-Presenters: Abhyuthan Srivastava
Abstract
Muscle contraction relies on interactions between actin filaments and myosin motors, but these interactions are regulated by additional structural proteins within the cytoskeleton. One such protein, α-actinin-2, crosslinks actin filaments and helps organize muscle architecture. Small chemical modifications such as phosphorylation may alter the structure of α-actinin-2 and influence how strongly it binds to actin. This project investigates whether phosphorylation-like mutations in the actin-binding domain of α-actinin-2 change the speed at which actin filaments move across myosin motors.
To address this question, computational modeling will predict structural changes in α-actinin-2 and estimate its binding strength to actin. Experimentally, purified actin and myosin from porcine cardiac tissue and recombinant α-actinin-2 proteins will be tested using a Total Internal Reflection Fluorescence (TIRF) in vitro motility assay. By measuring actin filament sliding velocities under different conditions, we aim to determine whether phosphorylation strengthens actin binding and increases frictional resistance. These findings will provide new insight into how cytoskeletal proteins regulate muscle mechanics.
Keywords: Heart, Cardiology, Muscle