Research Symposium
26th annual Undergraduate Research Symposium, April 1, 2026
Blake Roberts Poster Session 2: 10:45 am - 11:45 am / Poster #302

BIO
Blake Roberts is a third-year biological science major at Florida State University. Since October of 2025, he has assisted in Dr. Qian Yin's immunology lab at the Institute of Molecular Biophysics, where he spends 20-30 hours per week utilizing E. coli to express human guanylate binding proteins and purifying them for structural and functional analysis. He also has assisted Dr. Jeremy Owen's trace metal geochemical lab at Earth, Ocean, and Atmospheric Sciences where he purifies rock samples and prepares them for isotope and composition analysis through Mass Spectrometry.
Blake plans to pursue immunology in graduate school and continue a career in academia to become a research professor at the university level.
Human Guanylate-Binding Protein 4: Purification, Functional Assay, and Protein Sequence Analysis
Authors: Blake Roberts, Taiwo Scholes AdewoleStudent Major: Biological Science
Mentor: Taiwo Scholes Adewole
Mentor's Department: Biological Science Mentor's College: College of Arts and Sciences Co-Presenters:
Abstract
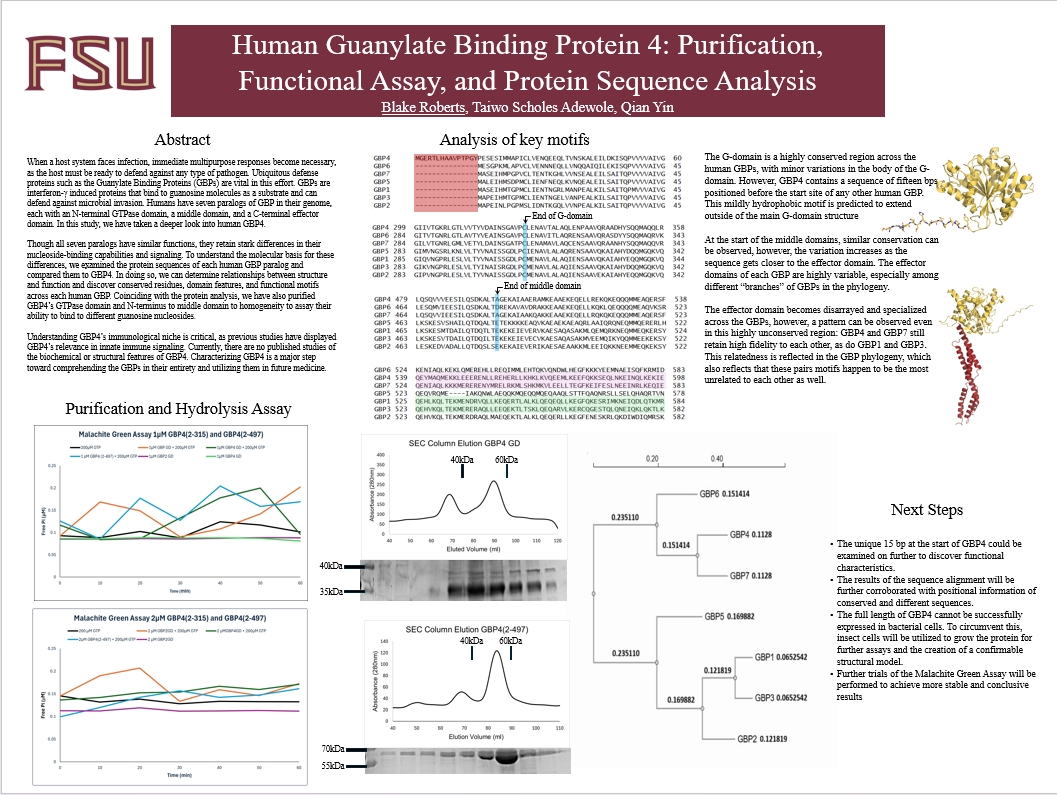
When a host system faces infection, immediate multipurpose responses become necessary, as the host must be ready to defend against any type of pathogen. Ubiquitous defense proteins such as the Guanylate Binding Proteins (GBPs) are vital in this effort. GBPs are interferon-γ induced proteins that bind to guanosine molecules as a substrate and can defend against microbial invasion. Humans have seven paralogs of GBP in their genome, each with an N-terminal GTPase domain, a middle domain, and a C-terminal effector domain. In this study, we have taken a deeper look into human GBP4.
Though all seven paralogs have similar functions, they retain stark differences in their nucleoside-binding capabilities and signaling. To understand the molecular basis for these differences, we examined the protein sequences of each human GBP paralog and compared them to GBP4. In doing so, we can determine relationships between structure and function and discover conserved residues, domain features, and functional motifs across each human GBP. Coinciding with the protein analysis, we have also purified GBP4’s GTPase domain and N-terminus to middle domain to homogeneity to assay their ability to bind to different guanosine nucleosides.
Understanding GBP4’s immunological niche is critical, as previous studies have displayed GBP4’s relevance in innate immune signaling. Currently, there are no published studies of the biochemical or structural features of GBP4. Characterizing GBP4 is a major step toward comprehending the GBPs in their entirety and utilizing them in future medicine.
Keywords: Immunology, Guanylate Binding Protein, Sequencing